Chemistry Research at UNE
UNE has a strong tradition of research and teaching in chemistry across the breadth of the discipline. We are proud that in the 2015 ERA (Excellence in Research Australia), chemistry at UNE was recognised as above world-class in macromolecular and materials chemistry.
Our Research
UNE researchers are active in research in both synthetic and natural macromolecules, with a focus on systems of relevance to industry and regional Australia. Active areas of research include the development of polymeric materials for drug delivery applications, 3D printing advanced materials for improving healthcare systems, fundamental studies to increase the versatility of controlled polymerisation, development of advanced soft materials through supramolecular self-assembly, and improving the physical properties of cereal foods. For current research opportunities contact Dr Ali Bagheri, Dr Brendan Wilkinson, Associate Professor Chris Fellows or Professor Trevor Brown. Natural products have provided many of the common drugs and drug classes used in humans including opioids for pain, anticancer agents as well as drugs to treat asthma. Natural Product isolation at UNE is focussed on the discovery of novel compounds from Australian native flora. These compounds are then used in bioactivity studies. Another research focus is the transformation of natural product resources including biomass into pharmaceuticals and compounds of industrial significance. For more information, contact Dr Ben Greatrex. The study of organic synthesis allows chemists to build complex molecules from simple and readily available precursors. Chemistry researchers at UNE are developing new ways to carry out reactions and synthesise challenging molecules with complex behaviour and potentially valuable biomedical applications. UNE has expertise specialising in carbohydrate synthesis, organometallic synthesis, green asymmetric synthesis targeted at chiral pharmaceuticals, and surfactant and supramolecular self-assembly processes. For current research opportunities contact Dr Ben Greatrex, Dr Michelle Taylor or Dr Brendan Wilkinson. Chemistry researchers at UNE are developing new biological redox sensors based upon N-heterocyclic carbene (NHC) macrocyclic metal complexes. This work requires quantitative elucidation of the relationships between structure and properties of the complexes so sensors can be “tuned” for specific applications. Further investigations are looking at different structural motifs (including macrocyclic and pincer complexes) and their applications as catalysts for a variety of reaction types. For current research opportunities, contact Dr Michelle Taylor or Dr Peter Lye. Molecular simulation and high-performance computing are used by researchers to gain an atomic level understanding of important chemical and biological systems. This assists in the development of new hypotheses and models which ultimately help to address important questions and develop innovative solutions in many areas of applied science. Researchers at UNE are using such methods to inform the design of several applications, such as polymers with greater efficiency in inhibiting industrial scale formation and an economically and environmentally viable coating to reduce evaporation from water storage bodies. For current research opportunities contact Dr Erica Smith. Chemistry researchers at UNE are developing new analytical techniques to study both industrial processes and our changing environment. Areas of active research include the investigation of metal ions in wine and their role in the production of unpleasant characteristics through complex formation with organic acids, the study of scale growth inhibition of common scale forming species like calcium carbonate and calcium oxalate in the presence of polyacrylic acids using both optical and conductivity sensors and the development of disposable sensors for biomedical devices. The application of these analytical methodologies to environmental systems is also allowing UNE chemists to address concerns facing Australia and the world. Research projects include the development of deployable devices to monitor the impact of climate change on the oceans and sensors to measure real-time calcification in coastal ecosystems. For current research opportunities, contact Dr Peter Lye. Chemistry at UNE has a long history of excellence in teaching and this includes an active interest in the scholarship of teaching and learning in chemistry. Research in this area focusses on online and distance learning, pedagogical content knowledge (PCK) in chemistry, e-learning and regional and rural higher education. For current research opportunities contact Dr Erica Smith.
We offer a Royal Australian Chemical Institute (RACI) accredited undergraduate major in chemistry and also medicinal chemistry. Undergraduates can engage in research by taking a dedicated research unit (SCI395) and will also experience research modules embedded within later year units. Students can undertake PhD and MSc postgraduate research degrees in areas supported by our academic staff. More information on the chemistry teaching program can be found here.
Research in the Spotlight
Copper complexes as a source of redox active MRI contrast This study demonstrates the potential for copper complexes to be used as redox sensitive MRI contrast agents for the detection of oxidative stress. Although not as effective as lanthanides in terms of contrast ability, the reversible redox properties and ability to turn-on contrast in the presence of appropriate oxidants highlights the opportunities copper complexes offer for redox sensitive imaging. Robust and Ultrasensitive Polymer Membrane-Based Carbonate-Selective Electrodes This paper describes a general method to improve the limit of detection of ion-selective electrodes by up to four orders of magnitude. This approach was demonstrated with a range of ISEs, including seawater carbonate sensors. Palladium-Catalyzed Suzuki–Miyaura, Heck and Hydroarylation Reactions on (–)-Levoglucosenone and Application to the Synthesis of Chiral γ-Butyrolactones This article describes new ways to derivatise levoglucosenone, a readily available chiral synthon which is available in bulk from the pyrolysis of lignocellulose. The derivatives produced will be used in the synthesis of chiral pharmaceuticals and industrial chemicals. Inhibition of Homogeneous Formation of Magnesium Hydroxide Magnesium hydroxide is a major component of the alkaline scale formed in thermal desalination processes. This work shows that this scaling can be inhibited using low molar mass poly(acrylic acid) with hydrophobic end-groups, with a higher degree of hydrophobicity giving optimum inhibition than was previously HERON reactions of anomeric amides: understanding the driving force The HERON (Heteroatom Rearrangement On Nitrogen) reaction has been observed only in select anomeric amides; amides possessing low amide resonance, bearing two electronegative heteroatoms at the amide nitrogen. In this paper, using density functional theory, we demonstrate how the activation barrier for the HERON reaction can be separated into two components, the energy required to overcome amide resonance and the energy required for rearrangement, and that without the reduction of the amide resonance component, the activation barrier for the HERON reaction remains prohibitively large. The effect of Z-group modification on the RAFT N-vinylpyrrolidone, a ‘Less Active Monomer’, is difficult to polymerise controllably into block copolymers with ‘More Active Monomers’. This work demonstrates that N-pyridyl functionalised dithiocarbamates bearing electron donating Z substituents can easily homo- and copolymerise
agents
Biometals, 2015, 28(5), 903-912
DOI 10.1007/s10534-015-9875-3
L. Dunbar, R. J. Sowden, K. D. Trotter, M. K. Taylor, D.
Smith, A. R. Kennedy, J. Reglinski, and C. M. Spickett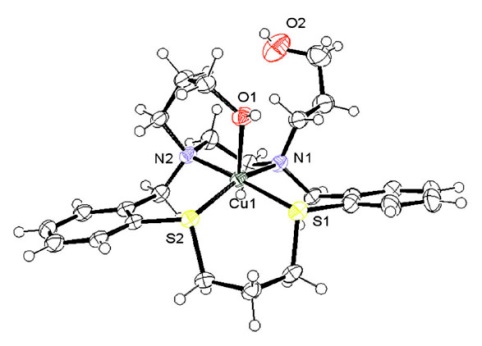
Analytical Chemistry, 2015, 87, 7515−7518
DOI: 10.1021/acs.analchem.5b01756
L. Mendecki, T. Fayose, K. A. Stockmal, J. Wei, S. Granados-Focil, C. M. McGraw and Aleksandar Radu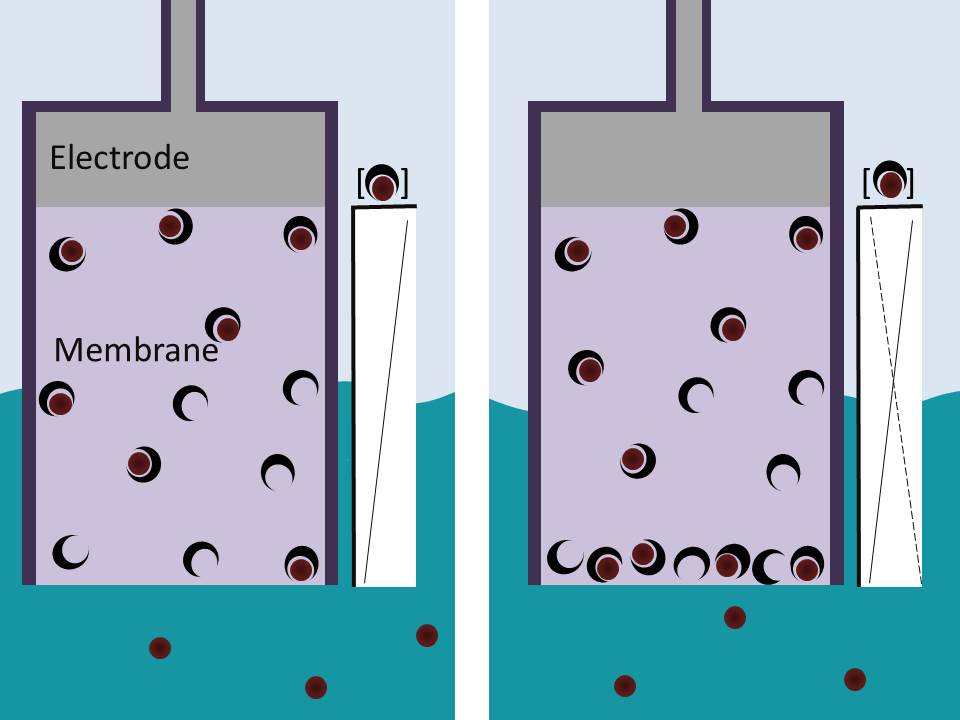
European Journal of Organic Chemistry, 2015, 2015(32), 6999-7008
K. P. Stockton, C. J. Merritt, C.J. Sumby and B. W. Greatrex
by Low Molar Mass Poly(Acrylic Acid) with Different End-Groups
Industrial and Engineering Chemistry Research, 2015, 54(7),
2201-2207
A. A. Al-Hamzah, E. J. Smith and C. M. Fellows
observed for calcium carbonate and calcium oxalate scales, giving insight towards the design of improved scale-inhibitors for thermal desalination plants.
Journal of Physical Organic Chemistry, 2015, 28 (3), 215-222
S. A. Glover and A. A. Rosser
polymerization of N-vinylpyrrolidone controlled by “switchable”
N-pyridyl-functional dithiocarbamates
Polymer Chemistry, 2015, 6, 7119-7126
S. J. Stace, G. Moad, C. M. Fellows and D. J. Keddie
N-vinylpyrrolidone with good control of dispersity, providing an effective new strategy for novel polymer materials.
Research Staff in Chemistry
- Dr Ali Bagheri (Polymer Chemistry)
- Professor Trevor Brown (Catalysis and green chemistry)
- Dr Siew Chong (Organometallic)
- Associate Professor Chris Fellows (Polymers and materials)
- Adjunct Professor Steven Glover (Physical organic)
- Dr Ben Greatrex (Organic/medicinal)
- Professor Amir Karton (Computational Chemistry)
- Dr Peter Lye (Coordination chemistry)
- Dr Adam Rosser (Organic)
- Dr Erica Smith (Molecular dynamics)
- Dr Michelle Taylor (Organometallic)
- Dr Brendan Wilkinson (Organic/bioorganic)
Research Facilities
The discipline is supported with state-of-the-art chemical analysis and characterisation tools. Undergraduate students are introduced to selected research equipment while postgraduate students are trained to use all relevant instruments as part of their degrees.
View the list of instrumentation available at UNE.

